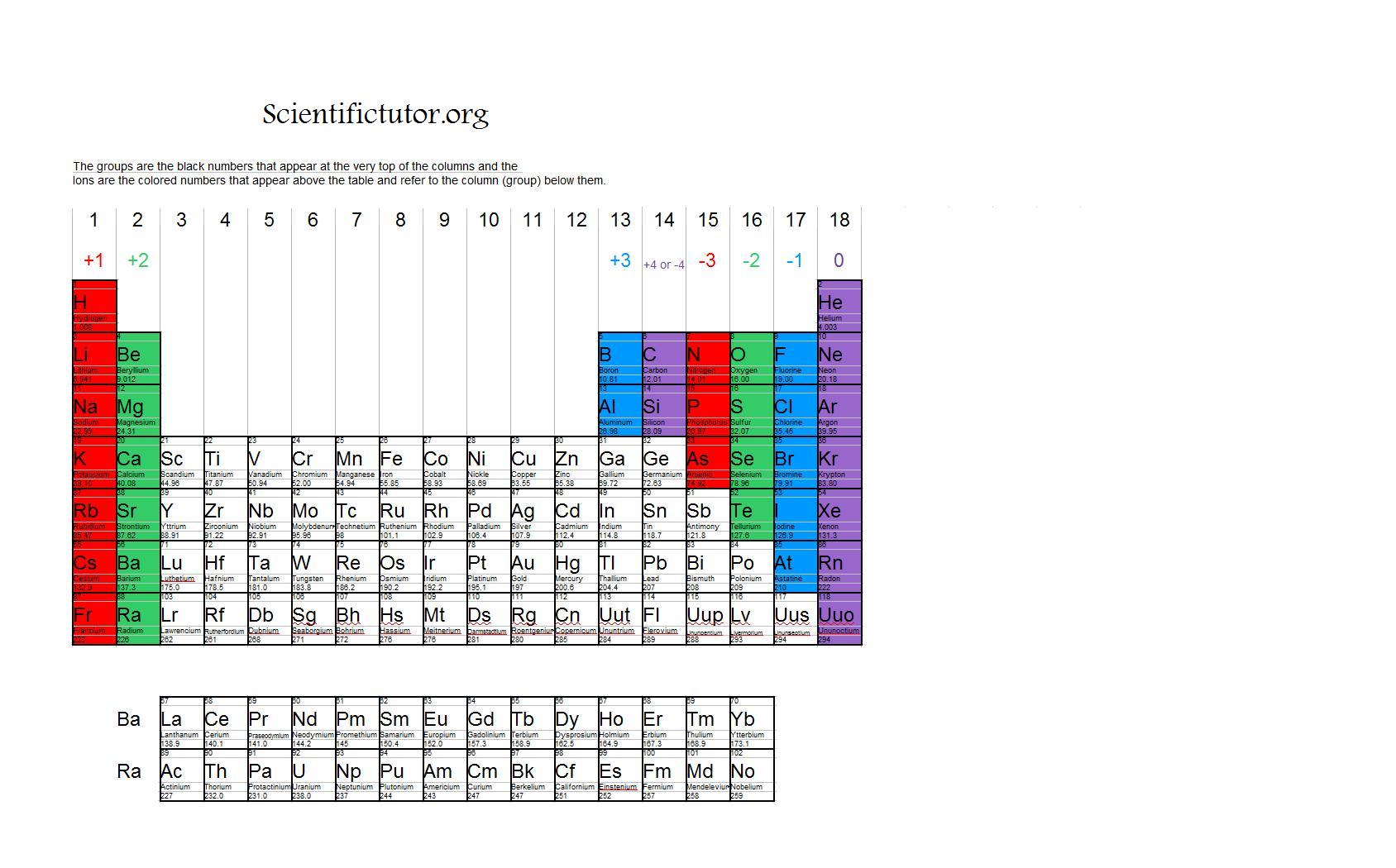
E) none of the above (A, B, or C) occurs. The number of dots equals the number of valence electrons in the atom. D) each of the above (A, B, and C) occurs. A Lewis electron-dot symbol (or electron-dot symbol or a Lewis symbol) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Neutron number plus atomic number equals atomic mass number: N ZA. Youve lost a whole layer of electrons, and the remaining 10 electrons are. 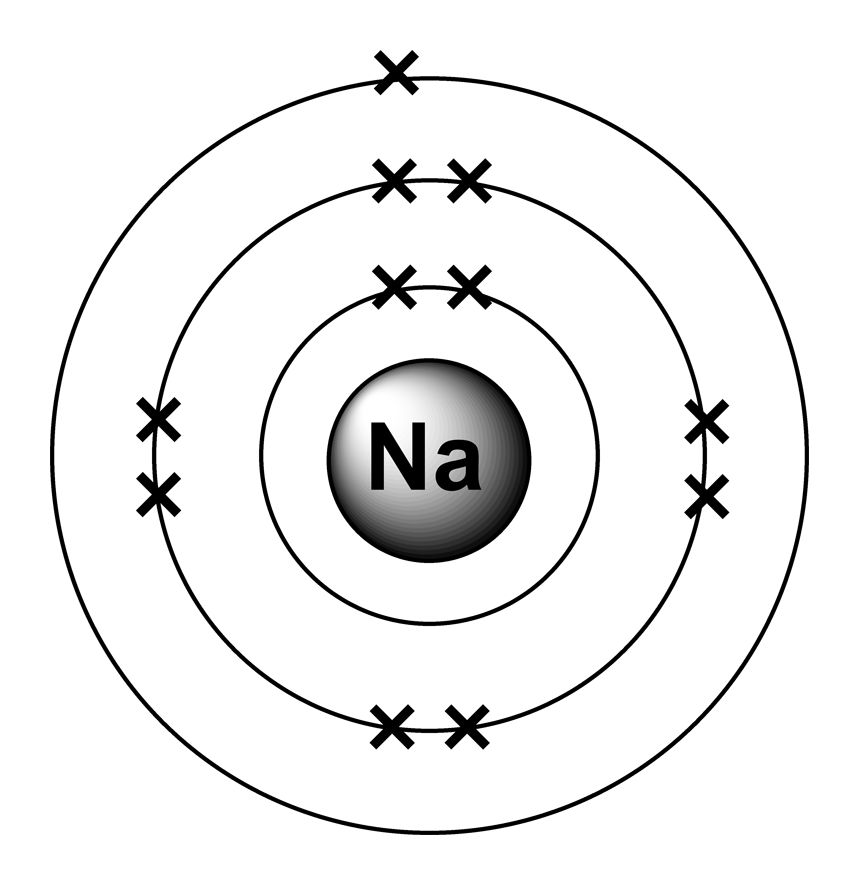
Although they are composed of ions, ionic compounds are: a. Positive ions are smaller than the atoms they come from. a compound composed of anions and cations. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. all halogen atoms have valence electrons and need to gain electron/s to achieve the configuration of a noble gas. C) the solvent-solute attractive forces overcome the solute-solute attractions. Mass numbers of typical isotopes of Sodium are 23. B) the negative end of water dipoles attract the positive ions. Note that not all of the protons and neutrons are visible. ( A) Hydrogen atom (atomic number 1, 1 proton, 1 electron) ( B) Oxygen atom (atomic number 8, 8 protons, 8 electrons). Therefore, a sodium atom has eleven protons and eleven electrons. Atoms prefer to gain the stability of octet, by having eight electrons in the outer shell, the electrons of the s and p orbitals. In this configuration we note that there is only one electron in the 3rd energy level. The atomic number of an element is equal to the number of protons and electrons in that element. The electron configuration of a neutral sodium atom is 1s22s22p63s1. Sodium is the 11th element of the periodic table so its atomic number is 11. By going through the periodic table, we see that the electron-dot symbol of atoms will never have more than eight dots around the atomic symbol.\): Naming the \(FeCl_2\) and \(FeCl_3\) Compounds in the Old/Common System. When an ionic compound dissolves in water: A) the positive end of water dipoles attract the negative ions. Sodium (Na), with atomic number eleven, has two electrons in shell 1, eight electrons in shell 2, and one electron in shell 3. Sodium is a classified alkali metal element and its symbol is Na. In ionic compounds, the charges of constituent ions must balance. When it forms the nitride ion, it gains three electrons to form a '3-' ion: 'N'(3-). Nitrogen is an element in group 5 of the periodic table. As such, it will form an electron by losing a single electron to form a '1 ' ion: 'Na' . Appearance: white powder, fragrance-free. 'Na'3'N' Sodium is an element in group 1 of the periodic table. Chemical Characteristics: Low solubility in water. Lithium Carbonate (Li2CO3) Uses: drug development. All except Lithium are soluble in water and stable to heat. The octet rule is a result of trends in energies and. It turns out that the Na ion has a complete octet in its new valence shell, the n 2 shell, which satisfies the octet rule. 
With the next element, sodium, the process starts over with a single electron because sodium has a single electron in its highest-numbered shell, the n = 3 shell. Group 1: Alkali Metals and Carbonates (X 2 CO 3) is the reaction between Li, Na, K, Rb and Cs with CO 3. 4.5: Elements: Defined by Their Numbers of Protons 4.6: Looking for Patterns: The Periodic Law and the Periodic Table 4.7: Ions: Losing and Gaining Electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
